In my previous blog post, I tore down the Kentli PH5 battery – a Li-ion battery that has an internal 1.5-volt regulator that allows for terrific voltage stability… up to a point. In terms of data collection, so far I have collected 55+ runs of data logs (248 MB of text files!) and still do not quite have all the data I want. As for the data that I do have, I will be disseminating them with as much thoroughness as possible.
Updated (May 1, 2018):
The battery’s self-discharge rate experiment has come to its conclusion – click here to read it.
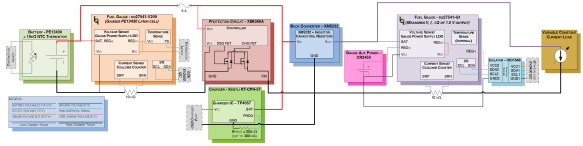
I forgot to add a diagram of my test setup – here’s a Visio diagram of the hardware used to test the battery’s performance… Click here to see the full-sized diagram.
Voltage vs. load current
As expected, the voltage output of the PH5 remains quite stable, up until roughly 2.1 amps where the voltage sags noticeably until the regulator goes into overcurrent protection mode.
A maximum load capacity of 2.1 amps seems to be a bit… limiting. That said, I have not done tests on the PH5’s transient load capacity, as it would require more automated control than what I currently have available.
Another issue with having such a flat discharge curve is that any device that performs fuel gauging using voltage alone will report 100% capacity, until it suddenly shuts down. This could be a big problem for digital camera users, as they will have no indication that their batteries are running low, until the device abruptly stops working. If the camera was writing an image to its memory card when the battery died, it could cause the image to be corrupted, or worse, damage the file system on the card!
Voltage vs. state-of-charge
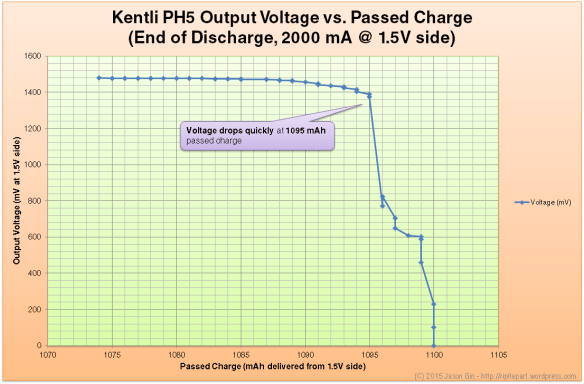
Unless you are running the battery at a high discharge rate, the output voltage will be flat at 1.5 volts before abruptly brickwalling and dropping to zero immediately at the end of discharge. At a high load (in the case of the graph below, at 2 amps), the voltage remains flat until the very end of the discharge cycle (99% depth of discharge for my test run), where it quickly tapers off and drops to zero.
Capacity vs. load
This is the big one, and it took a lot of work to get this data, especially at low loads (48+ hours of continuous logging is just asking for Murphy’s Law to come into play). I used almost 50 discharge runs to create the graph below.
This is where things get… interesting. I was expecting the capacity to peak at low currents then taper off as the load current increases. Instead, I noticed a definite ‘hump’ in capacity around the 250 mA mark (reaching a maximum of 1700 mAh / 2550 mWh), and only after that point did I see the expected downward slope in capacity, reaching 1200 mAh (1800 mWh) at the 2 amp mark.
This data brings forth some very interesting conclusions. The PH5’s capacity is inferior to its Ni-MH counterparts (even the relatively crappy ones), and at higher discharge rates it has similar capacity to that of an alkaline at the same load, albeit with much better voltage stability than the Ni-MH or alkaline chemistries.
Other findings
Although I won’t go into too much detail for the next few points (I haven’t gotten quite enough data to be presentable), there are some other issues with the battery that I think should still be mentioned.
One issue is the amount of heat the battery gives off at high loads. At 2.1 amps, I had to use a fan to blow cool air onto the DC-DC converter just to prevent it from entering its over-temperature shutdown mode. Although the converter itself can tolerate elevated temperatures, the Li-ion cell inside will not; the uneven heating that the cell will encounter could potentially degrade its lifespan in the long run.
Another problem is efficiency. At 1 amp, the DC-DC converter is about 75% efficient, and is only 65% efficient at 2 amps. I have not tested the converter’s efficiency at lower loads yet, but I doubt it will achieve more than 85-90% efficiency.
A potential issue with this battery is self-discharge. The buck converter remains active all the time, unless the converter or the Li-ion protection circuit enters a protective shutdown state. I have not had a chance to fully charge an unmodified battery in order to perform a long-term self-discharge test, but I will create another blog post for that, if/when the time comes. Update (May 3, 2018): See the top of the page for the link to the self-discharge test results.
Conclusion
Overall, I’m on the fence when it comes to this battery. Its innovative design does provide unparalleled voltage stability, but its low capacity even at moderate discharge rates dampens the fun significantly. Additionally, the 2.1 amp discharge limit could prove to be a bottleneck for some high-drain applications; this, coupled with the cell’s tendency to shut down abruptly when the internal cell runs empty could potentially cause file system corruption for digital cameras that have not been designed to handle such sudden power interruptions.
Also, the batteries are very costly. At about $10 per cell, you may want to think twice about replacing all your current disposable and rechargeable batteries with these newfangled Li-ion ones. Don’t forget the charger either, as a special charger is required to make contact with a recessed terminal on the top of the battery.
Overall, this cell is… interesting. Just don’t expect a miracle in a steel can.
Pros:
- Excellent voltage stability, even at high loads
- Li-ion chemistry allows for a very lightweight cell, even with the addition of a DC-DC converter
- High output voltage could allow some devices to run more efficiently
Cons:
- Low capacity – provides a mere 1200 mAh (1800 mWh) @ 2 amps, and up to 1700 mAh (2550 mWh) @ 250 mA (even alkaline batteries can do better than this)
- Abrupt shutdown when the battery is overloaded, overheated, or over-discharged
- Runs hot at high loads (and therefore is fairly inefficient)
- 1.5 MHz converter and unshielded inductor can cause excessive EMI (electromagnetic interference) in sensitive devices
- Expensive! Costs approximately $10/cell
- Requires proprietary charger
Bottom Line: This is a niche product and should not be considered a universal replacement for alkaline or Ni-MH AA batteries.



Pingback: Teardown of Kentli PH5 1.5 V Li-Ion AA battery | Rip It Apart – Jason's electronics blog-thingy
Super impressive review. Which rechargeable AA and AAA batteries you recommend to purchase ? What items can take only 1.2V or 1.25V rechargeable batteries ? .Some can only take a 1.5V battery. Guessing only flash lights and camera flashes can take the 1.2V rechargeables.
LikeLike
Pingback: Self-discharge test of Kentli PH5 1.5V Li-ion AA (Part 1) | Rip It Apart – Jason's electronics blog-thingy
Neat review. These are the curves that I wish that had come with the documentation.
I use a pair of Bose QC15s pretty much only when I travel, so I’d prefer not to have to deal with replacing battiers. Unfortunately they don’t play well with NiMH rechargeable batteries because they drop out somewhere between 1.1V and 1.2V. Practically speaking, this means that they can only be used for about three hours whereas Alkaline batteries last closer to thirty hours.
I’m hoping that the 1.5V output will let me operate my headphones for longer. The 20-30µA quiescent current is a little lame, but it’s not too hard to charge them overnight before a trip.
LikeLike
I feel like you are forgetting the most important pros and cons.
Pros compared to NiMH:
No memory effect
Little cycle loss
Cons compared to NiMH:
Calendar loss
The amount of calendar loss would really be much more interesting to know than the self-discharge rate (as it’s probably much higher)
LikeLiked by 1 person
Interesting review! These cells perhaps suit my need very well, that neither Alkaline nor NiMH rechargeable battery can do.
I am using two techlite TE116 LED flashlight on my mountain bike. They offer promising illumination on extensive longitudinal and lateral range with 3xAAA. They require total voltage supply to be above a threshold at around 4V. Below that point, the luminance drops like crazy. Alkaline battery roughly sustain 3 to 4 hours use before dropping below 1.3V each, while NiMH ones would never work. 6xAAA alkaline battery are dropped out from my flashlight every two nights of biking. They still work on all kind of devices but not my flashlight.
The LED of the flashlight should be rated less than 3Watt. Current draw should be well below 2Amp. I don’t mind charging the battery every night as long as they barely last two hours before starving. I can pack 3 more alkaline AAA for emergency.
Let’s see if it works.
LikeLike
Some updates:
Checked the specification of the LED of my flashlight, Cree XP-E 150 Lumen, current draw 1-1.5A, operable voltage around 2.8-3.4V. Turn out it wasn’t the problem of voltage drop, it is about current drop. GP ultra alkaline cells can provide ~1.4V and >1A supply for 2 to 3 hours, drop to ~1.2V 0.7A and fail to support the flashlight.
Got a few eneloop pro AAA rated at 950mAh. They provide consistent supply at 1.2V 1.4A about 45mins before dropping dead. So… the ~600mAh Kentli cells are out of question. Their capacity is way way too low.
LikeLike
Max,
Check these LED flashlights on Amazon.
They both use either a rechargeable battery or 3 AAA cells. Rechargeable:powered by 1 x 18650 3.6v/3.7v battery(not included ) or 3X AAA battery( not included)
I own both of these and in spite of the different lumen rating there is no real difference in brightness of the beam.
These things are cheap!! And there are many brands selling the identical light with a different name. I bought some of them too.
Use the 18650 battery and get a charger for it. It works well. All these lights have a reduced intensity mode which might allow for longer operation. I haven’t tried that. They also have a beam size adjustment allowing you to go from a very small spot beam to a very wide one.
I love these things and bought several to have around the house and in the car. The Li Ion battery has a long shelf life so it’s ready when you need it.
LikeLike
Thanks! I’m interested to see that AA lithium is available at all as there were significant reasons why it was not possible to get these.
1200mAh low drain is still better than the cheap “3000mAh!!!” junk being shipped with solar lights, verified capacity on these to be less than 700mAh and in some cases lower still.
Might get some just to retrofit solar garden lights as this would be doable.
LikeLike
Jason, thank you very much for this review, it is incredibly helpful!
I am researching battery capacity for a personal electronics projects and your post as proven extremely valuable.
LikeLike
I really can’t find the decent words to thank you as you deserve for this great test, it helps me to decide which batteries I should buy for my sound recorder, I was expecting buying these instead of Panasaonic eneloop pro. Really thank you
LikeLike
Did I miss 2here you tested how long they hold a charge without use? Seems like the most important thing I want to know, but I don’t recall seeing it.
LikeLike
It’s a test that’s still underway. I haven’t made a recent update on this, but hopefully I can make another blog post on it.
Jason
LikeLike
Pingback: Harnessing the power of lithium polymer in a 1.5 V AA package | Just Novel Gadgets
Pingback: Lithium Polymer Technology inside of a 1.5V AA Battery | Just Novel Gadgets
I sent an E-Mail to unbatteries.com and referenced your Web site. They were very prompt in replying. I have attached their comment and contact information.
Your comment would be appreciated.
From
To
Joseph Moskwa
Message body
Hello,
Thanks for your messages.
About question “the output voltage will be flat at 1.5 volts before abruptly brickwalling and dropping to zero immediately at the end of discharge” that is right for model PH5, we have another models PK5 and PM5 they are descending of voltage at the end of discharge.
About compare with Alkaline and Ni-MH that are to much advantages like charging time , memory effect, weitht ,lifetime and so on.
LikeLike
Hi Joseph,
Thanks for forwarding the manufacturer’s messages to me. I’ll definitely have to look into their other models to see how they perform. Although it doesn’t address the capacity concerns, their light weight is a definite advantage compared to Ni-based chemistries.
A quick Google search turned up one of their product briefs that outlines the difference between their product lines. It looks like the PK5 and PM5 have reduced maximum output current (1.5 A).
I might try contacting the manufacturer and asking if they’d be interested in sending me a selection of their other models for review – it could be interesting.
Thanks,
Jason
LikeLike
So are the pK5 and PM5 batteries better than ph5? Should I be looking to buy them instead of ph5. Are they a newer model? Could you please help on deciding which battery is best to buy as far as it goes for lithium batteries.
I was thinking of using them on my front door lock to enter the house.
Thank you.
Tom.
LikeLike
Thank you very much for doing this. Very helpful.
LikeLike
Excellent detailed review.
Will you review Irevoor rechargeable batteries also?
LikeLike
I didn’t originally have plans to, so thanks for sharing that link. Looks promising – good thing I have Amazon Prime!
Thanks,
Jason
LikeLike
Do you think these would be suitable for a game camera? I have 3 cellular cameras… one of the biggest issues is when the batteries start getting low, they become intermittent, the do not send any pictures after a while… the idea with a cellular game camera is to not have to go to it to replace batteries, but rather get the pictures sent to you. So, I would actually prefer batteries that shut off suddenly… I can check the web page to see if the camera is responding. If not, then time to charge the batteries… These game cameras take 12 batteries each, so the investment would be significant. But every year I am spending a LOT on Energizer Lithium disposables. The only thing I would worry about is heat… the cameras would already be out in the hot Texas weather. I don’t think these cameras have much of a current draw however.
So what would be your thoughts on this? Thanks in advance
PJ
LikeLike
Hi Phil,
At 12 batteries per unit, times 3… that’s a pretty large number of batteries you’re running through. How long do your Energizer lithium batteries last?
What model of cellular game cameras do you have? I can’t say too much on how well these Kentli batteries would work for your application, especially taking outdoor daytime heat into consideration. Cellular data transmission is pretty power-intensive, even if the average power draw is fairly low (it doesn’t help that the power used for transmission is very “bursty” which makes it harder to predict how well a battery with built-in DC-DC conversion will perform). That said, have you looked into Ni-MH rechargeable batteries? It may be possible to try some reputable LSD (low self-discharge) batteries from, say, Eneloop, but I suspect that the cameras may not be designed for the lower terminal voltage of Ni-based chemistries.
Regards,
Jason
LikeLike
Thanks for the review. I would like to know a bit more about EMI issues. Have you done any test on this aspect and had some related figures? Or have you (correctly) pointed out this potential problem as you read somewhere that these batteries have 1.5 MHz converter and unshielded inductor?
LikeLike
My statements on potential EMI problems were based on my observations that the inductor was unshielded, and the buck converter’s frequency was obtained from its datasheet.
I have not done any oscilloscope-based measurements (ripple, spectrum, etc.) as I did not even own a (digital) oscilloscope until roughly a month ago. I may revisit this subject and see the actual output characteristics of the converter.
Jason
LikeLike
Thanks for the reply Jason. EMIs are our main concern, therefore what you have described represents a deterrent to use them.
Massimo
LikeLike
Pingback: Completed: Self-discharge test of Kentli PH5 1.5V Li-ion AA (Part 6) | Rip It Apart – Jason's electronics blog-thingy
Your article was a refreshing uptick in engineering competence. I was charmed by the 1.5 volt buck converter trick. Under the assumption that my Garmin 600 GPS was designed for 1.5 volt alkaline, 1.5 volt Li-ion seemed the thing to purchase. Wrong. As you point out, most devices are designed to run at the 1.2 volt plateau that alkaline batteries quickly establish under use, also where NiMH operates. Converting WAH to MAH and noting the 2x price advantage made the choice of Eneloop Pro easy.
Thanks.
Chuck Tyler
LikeLike
Thanks for the kind words, Chuck – I really appreciate it!
Jason
LikeLike
DO NOT BUY!!!
I bought a pack of 4 of these off of ebay. After the first charge, 1 stopped working, would no longer hold charge. I just did a second charge and another battery stopped working. I am down to 2 batteries in less than a month.
LikeLike
I’ve purchased over 20 of them (aaa and aa), and I personally use 14.
no duds so far.
LikeLike
My Canon S2IS camera uses 4 AA batteries. I’ve been using NiMH rechargeables, but the camera shuts down and shows “Change the Batteries” around 4×1.28v, so I don’t get much mileage per recharge. Cleaning the terminals hasn’t helped. I’m trying an experiment: 2 NiMH and 2 1.5v Li-ion batteries (in series, of course). I hope to get the full mAh from the NiMH’s this way while only using 2 $10 batteries. Wish me luck!
LikeLike
I personally don’t recommend mixing battery types. Unpredictable results could occur, and it runs the risk of damaging the batteries if they end up reverse charging.
Still, could be an interesting experiment in the name of science!
LikeLiked by 1 person
Pingback: Ramble: 2018 in review | Rip It Apart – Jason's electronics blog-thingy
Very Nice Blog..!!
Thank You For sharing this information with us.
LikeLike
Pingback: Ramble: 2019 in review | Rip It Apart – Jason's electronics blog-thingy
Just found this review. Nice work.
The regulated rechargeable battery field has changed a lot in the lasat few years. Have you got plans to test the Jugee and other recent entries?
LikeLike
Unfortunately, rechargeable batteries haven’t piqued my interest as much lately. I might (re)visit them in the future but there’s no defined plans for them right now.
LikeLike
Pingback: Review of XTAR Li-ion 4150mWh AA / 1200mWh AAA batteries and L8 USB-C 8-bay charger | Rip It Apart – Jason's electronics blog-thingy