In my previous blog post, I tore down the Kentli PH5 battery – a Li-ion battery that has an internal 1.5-volt regulator that allows for terrific voltage stability… up to a point. In terms of data collection, so far I have collected 55+ runs of data logs (248 MB of text files!) and still do not quite have all the data I want. As for the data that I do have, I will be disseminating them with as much thoroughness as possible.
Updated (May 1, 2018):
The battery’s self-discharge rate experiment has come to its conclusion – click here to read it.
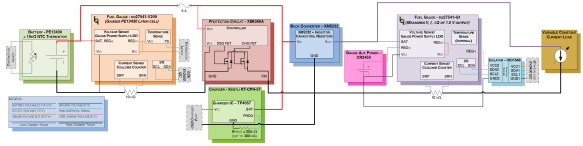
I forgot to add a diagram of my test setup – here’s a Visio diagram of the hardware used to test the battery’s performance… Click here to see the full-sized diagram.
Voltage vs. load current
As expected, the voltage output of the PH5 remains quite stable, up until roughly 2.1 amps where the voltage sags noticeably until the regulator goes into overcurrent protection mode.
A maximum load capacity of 2.1 amps seems to be a bit… limiting. That said, I have not done tests on the PH5’s transient load capacity, as it would require more automated control than what I currently have available.
Another issue with having such a flat discharge curve is that any device that performs fuel gauging using voltage alone will report 100% capacity, until it suddenly shuts down. This could be a big problem for digital camera users, as they will have no indication that their batteries are running low, until the device abruptly stops working. If the camera was writing an image to its memory card when the battery died, it could cause the image to be corrupted, or worse, damage the file system on the card!
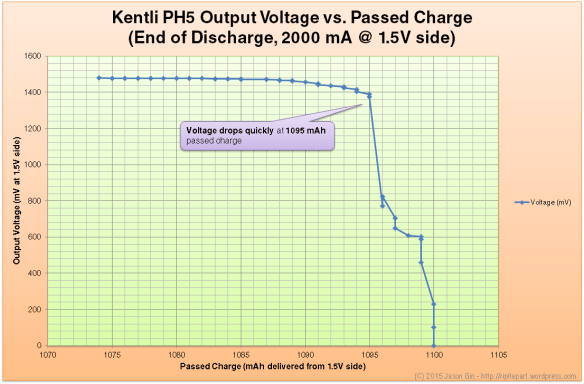
Voltage vs. state-of-charge
Unless you are running the battery at a high discharge rate, the output voltage will be flat at 1.5 volts before abruptly brickwalling and dropping to zero immediately at the end of discharge. At a high load (in the case of the graph below, at 2 amps), the voltage remains flat until the very end of the discharge cycle (99% depth of discharge for my test run), where it quickly tapers off and drops to zero.
Capacity vs. load
This is the big one, and it took a lot of work to get this data, especially at low loads (48+ hours of continuous logging is just asking for Murphy’s Law to come into play). I used almost 50 discharge runs to create the graph below.
This is where things get… interesting. I was expecting the capacity to peak at low currents then taper off as the load current increases. Instead, I noticed a definite ‘hump’ in capacity around the 250 mA mark (reaching a maximum of 1700 mAh / 2550 mWh), and only after that point did I see the expected downward slope in capacity, reaching 1200 mAh (1800 mWh) at the 2 amp mark.
This data brings forth some very interesting conclusions. The PH5’s capacity is inferior to its Ni-MH counterparts (even the relatively crappy ones), and at higher discharge rates it has similar capacity to that of an alkaline at the same load, albeit with much better voltage stability than the Ni-MH or alkaline chemistries.
Other findings
Although I won’t go into too much detail for the next few points (I haven’t gotten quite enough data to be presentable), there are some other issues with the battery that I think should still be mentioned.
One issue is the amount of heat the battery gives off at high loads. At 2.1 amps, I had to use a fan to blow cool air onto the DC-DC converter just to prevent it from entering its over-temperature shutdown mode. Although the converter itself can tolerate elevated temperatures, the Li-ion cell inside will not; the uneven heating that the cell will encounter could potentially degrade its lifespan in the long run.
Another problem is efficiency. At 1 amp, the DC-DC converter is about 75% efficient, and is only 65% efficient at 2 amps. I have not tested the converter’s efficiency at lower loads yet, but I doubt it will achieve more than 85-90% efficiency.
A potential issue with this battery is self-discharge. The buck converter remains active all the time, unless the converter or the Li-ion protection circuit enters a protective shutdown state. I have not had a chance to fully charge an unmodified battery in order to perform a long-term self-discharge test, but I will create another blog post for that, if/when the time comes. Update (May 3, 2018): See the top of the page for the link to the self-discharge test results.
Conclusion
Overall, I’m on the fence when it comes to this battery. Its innovative design does provide unparalleled voltage stability, but its low capacity even at moderate discharge rates dampens the fun significantly. Additionally, the 2.1 amp discharge limit could prove to be a bottleneck for some high-drain applications; this, coupled with the cell’s tendency to shut down abruptly when the internal cell runs empty could potentially cause file system corruption for digital cameras that have not been designed to handle such sudden power interruptions.
Also, the batteries are very costly. At about $10 per cell, you may want to think twice about replacing all your current disposable and rechargeable batteries with these newfangled Li-ion ones. Don’t forget the charger either, as a special charger is required to make contact with a recessed terminal on the top of the battery.
Overall, this cell is… interesting. Just don’t expect a miracle in a steel can.
Pros:
- Excellent voltage stability, even at high loads
- Li-ion chemistry allows for a very lightweight cell, even with the addition of a DC-DC converter
- High output voltage could allow some devices to run more efficiently
Cons:
- Low capacity – provides a mere 1200 mAh (1800 mWh) @ 2 amps, and up to 1700 mAh (2550 mWh) @ 250 mA (even alkaline batteries can do better than this)
- Abrupt shutdown when the battery is overloaded, overheated, or over-discharged
- Runs hot at high loads (and therefore is fairly inefficient)
- 1.5 MHz converter and unshielded inductor can cause excessive EMI (electromagnetic interference) in sensitive devices
- Expensive! Costs approximately $10/cell
- Requires proprietary charger
Bottom Line: This is a niche product and should not be considered a universal replacement for alkaline or Ni-MH AA batteries.